FDA warned Mars Petcare a roach infested pet food plant is a “regulatory violation”, but then the agency just walked away taking no enforcement action.
One year ago (7/11/2017 through 7/26/2017), the FDA inspected a Mars Petcare manufacturing facility in Columbus, Ohio as follow up to a Mars Cesar dog food recall that occured 9 months prior. The Cesar recall only covered two lot codes of the dog food. This pet food plant manufactured multiple brands of wet pet food for Mars; Pedigree, Cesar, Whiskas, Nutro, Iams.
The following are excerpts from the 2017 FDA Establishment Inspection Report of the Mars pet food facility. The report discloses the ugly truth in pet food; if you are a Big Dog in pet food, the FDA takes little to no regulatory action against you – no matter the violations you commit.
Bold added in quotations for emphasis.
Inspectional Observations
1. Failure to inspect, segregate, or otherwise handle raw materials and ingredients used in manufacturing under conditions that will protect the animal food against contamination and minimize deterioration.2. Failure to take effective measures to exclude pests from your plant and protect against contamination of animal food by pests (Discussion Item from 10/27/2016 EI).
The FDA inspection found that Mars Petcare ‘failed’ to “minimize deterioration” of pet food raw ingredients. This ‘failure’ was not specific to just one brand of pet food, the ‘failure’ was for the entire plant and all pet foods made there.
And the FDA inspection found Mars Petcare ‘failed’ to prevent the pet foods from being contaminated by pests. Again, this ‘failure’ was for the entire plant and all the pet foods manufactured there.
This Mars pet food plant had a significant problem with “German cockroach infestation” that was present in the 2016 inspection AND the 2017 inspection. During the previous FDA inspection of this Mars pet food plant (9 months earlier – October 2016), the FDA investigator “reviewed the firm’s pest control deviations – most notably the firm’s German cockroach infestation (which is most prominent on production lines).”
But, in the 2017 follow up inspection, FDA noted “a significant German cockroach infestation of the firm persists.“
The 2017 Mars inspection report included multiple pages of a “Pest Sighting Log” maintained by the pet food manufacturer. Below is one page. Take note of the “Comment(s) from Employee” dated 12/13/2017.
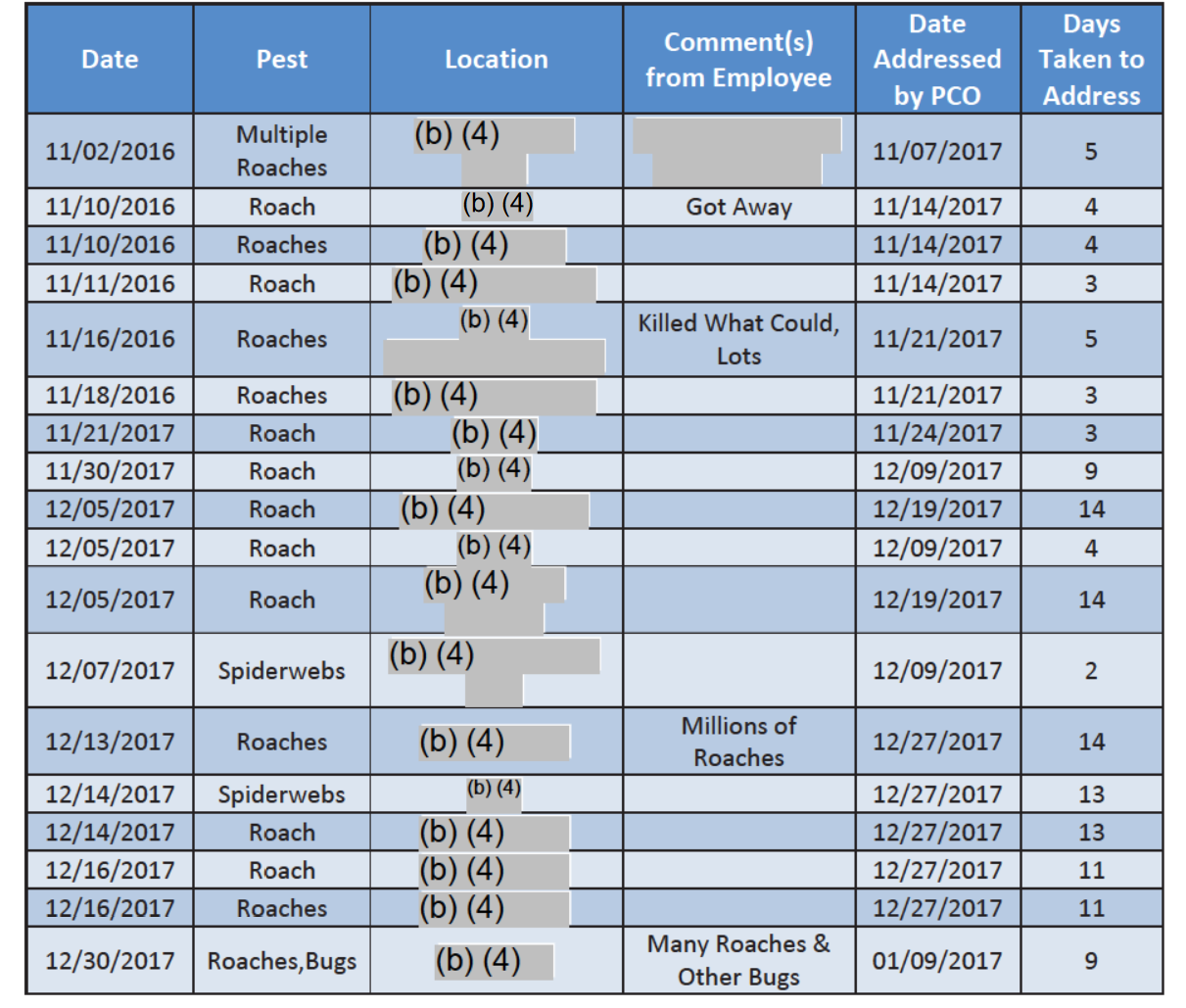
“Millions of Roaches.”
The inspection report stated: “The failure to take adequate measure to exclude and prevent pests from the manufacturing and related areas of the firm poses a significant public health safety concern while also being a regulatory violation.”
“A regulatory violation” – but…no recall occurred because of roach infestation. Five different brands of wet pet food are manufactured at this pet food plant (Pedigree, Cesar, Whiskas, Nutro, Iams). Again, not one product made in this plant that was documented by FDA to be infested with roaches, documented by FDA to be a “regulatory violation” was recalled.
Mars Petcare ignored the violation and warnings. FDA ignored the violation and allowed roach infested adulterated pet products to be sold to unknowing consumers.
And…Mars did not cooperate with FDA investigators during the inspection.
During the July 2017 inspection “Ms. Webster-Long led each day of the inspection and refused our requests for photocopies of documents citing corporate policy and instruction from the firm’s regulatory department. After each request for complaint logs and photocopies of documents, Ms. Webster-Long replied that either the regulatory team was not available, or the team refused the request. These numerous requests, delays in responses, and subsequent refusals delayed the completion of the inspection until 07/26/2017. Ms. Webster-Long led each of our walk-throughs of the manufacturing area, while often denying complete access to all areas of the firm. Most notably, Ms. Webster-Long appeared to intentionally avoid lines of the firm when we requested to observe those specific areas.
Besides denying FDA access to areas of the pet food plant the agency requested to visit, Mars also denied the FDA access to customer complaints.
“On 7/14/2017, following our repeated requests to review the firm’s consumer complaints – specifically those complaints involving the inclusion of FO’s (foreign objects) in the firm’s finished products – Ms. Webster-Long reported the Consumer Care Team in Franklin, TN, agreed to share their top three consumer complaint categories with us.”
Even though the FDA requested consumer complaints involving foreign objects in the pet foods, Mars instead provided the agency complaints on “Packaging, damaged or dented”, “Growth of mold”, and complaints regarding “Low meat/excess gravy or gel”.
The Mars managers interviewed by FDA assured the investigators there were no consumer complaints involving foreign objects in the pet food.
Ms. Webster-Long indicated the firm had not received any consumer complaints involving the white, plastic FO’s that were the concern for their previous, formal Class I Recall, initiated 10/07/2016.
Repeated verbal requests were made for the firm to share information they had regarding any FO’s – plastic or otherwise – the firm may have received. Ms. Webster-Long and Mr. Oskin refused to provide complaint information for FO inclusion in finished product, or complaints regarding adverse reactions occurring following exposure to the firm’s products.
But after FDA provided Mars Petcare with evidence of consumer complaints regarding foreign objects in the pet food, Mars employees changed their story…
During the inspection, we presented the firm with 13 consumer complaints reported to FDA, from 05/02/2016 to the present. After reviewing the information already available to Ms. Webster-Long; she confirmed she was aware of seven of the 13 consumer complaints FDA had received regarding products the firm manufactures. Of these seven consumer complaints, two involved white, plastic FO’s in the firm’s finished product; although Ms. Webster-Long and Mr. Oskin reported several times when questioned the firm had not received additional consumer complaints regarding white, plastic FO’s in the product they manufacture since they closed their investigation into the recall referenced above.
“During the inspection, we requested information from the firm for a consumer complaint received by FDA, on or about 11/06/2016, involving white plastic in the firm’s product. When questioned, Ms. Webster-Long reported the firm had also received this complaint and she initiated a formal PRIMP.”
PRIMP is a Mars Petcare internal acronym that stands for “Product Related Incident Management Process”. PRIMP is a Mars Petcare internal investigation of a consumer complaint regarding one of the pet foods made at the plant. Mars initially told the FDA they had NOT received consumer complaints regarding foreign objects in the pet food after the October 2016 recall. BUT, when FDA presented the pet food manufacturer with evidence, Mars admitted to receiving a consumer complaint AFTER the recall. In fact, Mars admitted “the firm is aware their incoming [redacted] utilized in the manufacture of many of the firm’s products, often contains plastic FO’s.” Mars admitted full knowledge of plastic foreign objects in raw materials used in the pet foods.
At the end of the FDA Inspection Report – under the section of “General Discussion With Management” is the following statement from FDA:
It was reiterated several times during the inspection the firm needs to share information requested by FDA during inspections. Refusing to provide requested records and information for review prevents FDA investigators from being able to thoroughly evaluate the firm’s manufacturing processes to ensure the safety of the firm’s products and determine compliance with applicable FDA law, rules, and regulations.
Consumers deserve to have a similar ‘General Discussion with FDA’. Consumers cannot trust the safety of pet foods when the FDA cowers to a pet food manufacturer. Why would FDA allow any company to refuse to cooperate during an inspection? Why wouldn’t FDA force a recall of every single pet food in that plant due to roach infestation that continued for at least 9 months?
Who is more at fault? Mars Petcare for ignoring law and safe manufacturing procedures or FDA for allowing adulterated pet food into the homes of unknowing consumers?
To read the full FDA Establishment Inspection Report of the Mars Petcare facility, Click Here.
Wishing you and your pet(s) the best,
Susan Thixton
Pet Food Safety Advocate
Author Buyer Beware, Co-Author Dinner PAWsible
TruthaboutPetFood.com
Association for Truth in Pet Food

Become a member of our pet food consumer Association. Association for Truth in Pet Food is a a stakeholder organization representing the voice of pet food consumers at AAFCO and with FDA. Your membership helps representatives attend meetings and voice consumer concerns with regulatory authorities. Click Here to learn more.
What’s in Your Pet’s Food?
Is your dog or cat eating risk ingredients? Chinese imports? Petsumer Report tells the ‘rest of the story’ on over 5,000 cat foods, dog foods, and pet treats. 30 Day Satisfaction Guarantee. Click Here to preview Petsumer Report. www.PetsumerReport.com

The 2018 List
Susan’s List of trusted pet foods. Click Here to learn more.
Have you read Buyer Beware? Click Here
Cooking pet food made easy, Dinner PAWsible
Find Healthy Pet Foods in Your Area Click Here



































Sally Roberts
July 21, 2018 at 1:36 pm
Totally disgusting but not surprised !! They feel they did there job going, warned them and that is it. Who suffers. the animals as usual !!
Casey Post
July 21, 2018 at 1:53 pm
So, who does the FDA serve? And why even bother inspecting if you’re not going to enforce?
landsharkinnc
July 21, 2018 at 2:37 pm
Our Government at work … as usual!
Ms. B Dawson
July 21, 2018 at 3:11 pm
From the Inspector’s comments: …”These numerous requests, delays in responses, and subsequent refusals delayed the completion of the inspection ….”
Maybe Independent pet food stores should employ these tactics when FDA officials show up unannounced and expect to abscond with hundreds of dollars of pet food vis-a-vis: “If FDA or State Comes Into Your Pet Food Store, Do You Know What Procedure Is?”.
Ian
July 21, 2018 at 3:14 pm
How can the manufacturer limit the inspection, make certain areas off limits, get caught in outright lies, and not suffer any penalty? Outrageous. Bravo to the employee who wrote “millions of roaches.” At least somebody with a shred of candor. The inspectors should be forced to eat or at least to feed their own pets the “food” manufactured at this facility.
Tom
July 21, 2018 at 7:13 pm
Because we keep electing senators who are bought and owned. Even worse, they publicly state their desire to deregulate.
Just remember: this is what a deregulated America looks like. Millions of roaches. Vote them out this November.
Jane Democracy
July 21, 2018 at 3:35 pm
I wish this was different in most kibble manufacturing facilities but unfortunately I know it’s not… Sigh
mickey
July 21, 2018 at 5:13 pm
how is the situation at the mars petcare plant today
Susan Thixton
July 21, 2018 at 5:27 pm
We don’t know if Mars has finally corrected the problem(s) or if conditions remain the same.
Teresa Johnson
July 21, 2018 at 7:04 pm
Mmmmm, makes me wonder what “secret ingredients” have crept into other Mars products?
If the plant is so overridden with roaches, you know some surely must have been transported to the distributors and retailers of their products. Makes me want to avoid any business carrying their products.
Cherry
January 19, 2021 at 12:51 pm
Yes!!!!!
Kim Nickell
July 22, 2018 at 12:37 am
I found a nail in a bag of pedigree dry dog good
Eve
July 23, 2018 at 12:38 am
Oh Kim I’m sorry you too found a hideous dangerous piece of metal in the pet food. At least you found it ‘before’ you fed your pet, I didn’t. My beautiful Silver-Point Persian had luckily left it on the plate inspecting it like ‘WTF?’ I ran over grabbing the plate and contacted the companu in distress. They tried to palm me off with a $10 voucher I refused so they gave me a few tins to replace it as they insisted and guess what…maggots this time I THREW ALL THE PET FOOD IN THE BIN and refused to feed my girl any more junk. From that point she only got human grade raw meat and raw meaty chicken necks/wings. This was 20 years ago. Who cares attitude is what these big pet food companies display. No amount of money could ever replace the lives and kinship our pets give us ever.
Kimberly
July 22, 2018 at 1:32 am
regardless…. if there roaches are gone – (doubtful) there are still nothing but human roaches who walk upright, all the way to the bank, with allllll off the *completely* UNINFORMED & UNEDUCATED & TRUSTING pet guardians hard earned money. WITH ZERO TRANSPARENCY THROUGHOUT. Thank you Ms Thixton – we’re who follow you and your tireless, thankless, infuriating, disappointing, & necessary work that you do. I admire your massive heart, spirit, fierce tenacity & your investigatory “chops”. I’ve been following you and Rodney Habib, Dr Becker, several other *industry-truthers* since my mother’s dog’s 4 of the 7 were POISONED & MURDERED due to greed & #profitsoverpets. The 4 died from eating New bag of Purina Beneful, Chicken, Healthy Weight… the other 3 dogs should never touch any Purina products – she fed them some other cheapskate garbage grocery store food, I believe it was Pedigree. (and they barely wanted to eat that). It was at that time I adopted my dogs. I didn’t know better and thought Nutro Max, Iams, Blue Buff, and Natures Variety. etc etc. Fast forward a year / half later, I finally was turned on to several safer kibble alternatives, the daily education I was providing myself – allowed me to get the big picture – I began weeding out anything with GRAIN, chicken. beef. white potato, *ANY* by products, tapioca. chickpeas. legumes (and all the 10 variations of the breakdown of peas…) *no corn* – no soy – no minerals from China – *no rice* – no dyes – no *natural flavoring added* – no BHA – NO ETHOXYQUIN. NO GMOS – NO HORMONES, OR ANTIBIOTICS ADDED TO THE MEAT – So, we are only dealing with environmental allergies with my Staffy Terrier whose seemingly allergic to everything. They aren’t eating raw yet, still too expensive. But with mommy SAFE kibble I still add, (organic as often as possible) : frozen broccoli spears are their favorite – cranberries, apples, squash, green beans occasionally, tomatoes, garlic granules, ACV, Krill oil, Sea Kelp, Coconut oil rotated with MCT Coconut oil, Raw Goat Milk Kefir, bee pollen, spirulina, Curcumin 95. Several other supplements that I can’t think of at this time. All that said.. no more allergies for my Border Collie / GSD but my Staffy Terrier is allergic to dust bunnies and a ton of pollens – during a windy day, she’ll be broken outin hives within 6 hours. USUALLY turning into a left ear yeast infection which i had cultured. This time… three ear had fungus, bacteria, AND Staph. This infection took a month and half to go away. Anyways… i’m learning all the time because of you specifically and the others I mentioned and the countless others I have not. (Ottowa Valley Dog Whisperer, Dr Karen Rosenfeld is AMAZING…. AHHHMAYZINGGG! *So my point, thank u for helping medo what’s right for my pets – my cats also have been upgraded to REALLY healthful food. I can’t begin to share the differences with them.. it’s truly miraculous. Thank you Ms. Thixton. We love you.
Kimberly
July 22, 2018 at 1:34 am
and I meantto say. regardless of processing plant passes inspection.. point being – companies like MARS should not even be on your radar to even think about purchasing from. Pet food or People food /products.. PERIOD
Joe Young
July 22, 2018 at 2:51 am
America has changed so much in the last decade. Buyer Beware! Stand up against deregulation. America sold out by lying elected officials. We need a list of politicians and their voting records before it’s too late.
Geneva Malone
July 22, 2018 at 9:16 am
I love my animals and Don’t trust these companies,Roaches carry Diseases that make Everyone and All Pets and People Sick and can Kill.
Peg
July 22, 2018 at 9:39 am
What frightens me, is the thought of them coming in with massive amounts of pesticides and not taking any precautions to protect the “feed” ingredients or the employees
Sheila E
July 22, 2018 at 12:45 pm
Didn’t even think about that – ack, you’re right!!
Eve
July 23, 2018 at 12:28 am
Well Said Susan. i LOVE this sinister music behind the snippet showing this is their full intentions on sneaking out kittylitterdoggydroppingspoofarthooharr (a polite way of saying sh$t) into our pet foods. y Every Pet Parent MUST take matters into their own hands NOW—seriously there’s non-stop evidence to say otherwise…Get Serious and only buy your pets RAW human-grade raw meat and raw meaty bones and/or include ONLY dry food that is purely dry-meat rolled into pieces – ziwi-peak is a brilliant alternative to the hooharr commercial pet foods stuffed with poisons toxins and garbage fillers. You’re wasting YOUR TIME and MONEY hap-haphazardly trusting the pet food industry. Its YOUR responsibility NOW so fight BACK and fight back HARD show them they have NO right to disrespect your best friend. Unless of course you don’t care about your pets being slowly poisoned and rotting from the inside out (of course I know you do-who wouldn’t – ummmm the majority of pet food companies that’s who).
Terri Christenson Janson
July 23, 2018 at 11:08 am
Yes, Peg, these were my very first thoughts. All the kibble will be exposed to the pesticides as well.
Jackie Hastings
July 24, 2018 at 4:55 pm
Why isn’t the media on this to alert the puplic
MARILYN JONES
February 5, 2019 at 7:33 pm
The Nutro treats that mars is the corporate office for gave all 3 of my dogs food poisoning. One almost died. Mars Pets refused to accept responsibility even after they were given the vet notes. And they refused to respond to the BBB. This company needs to be stopped!!